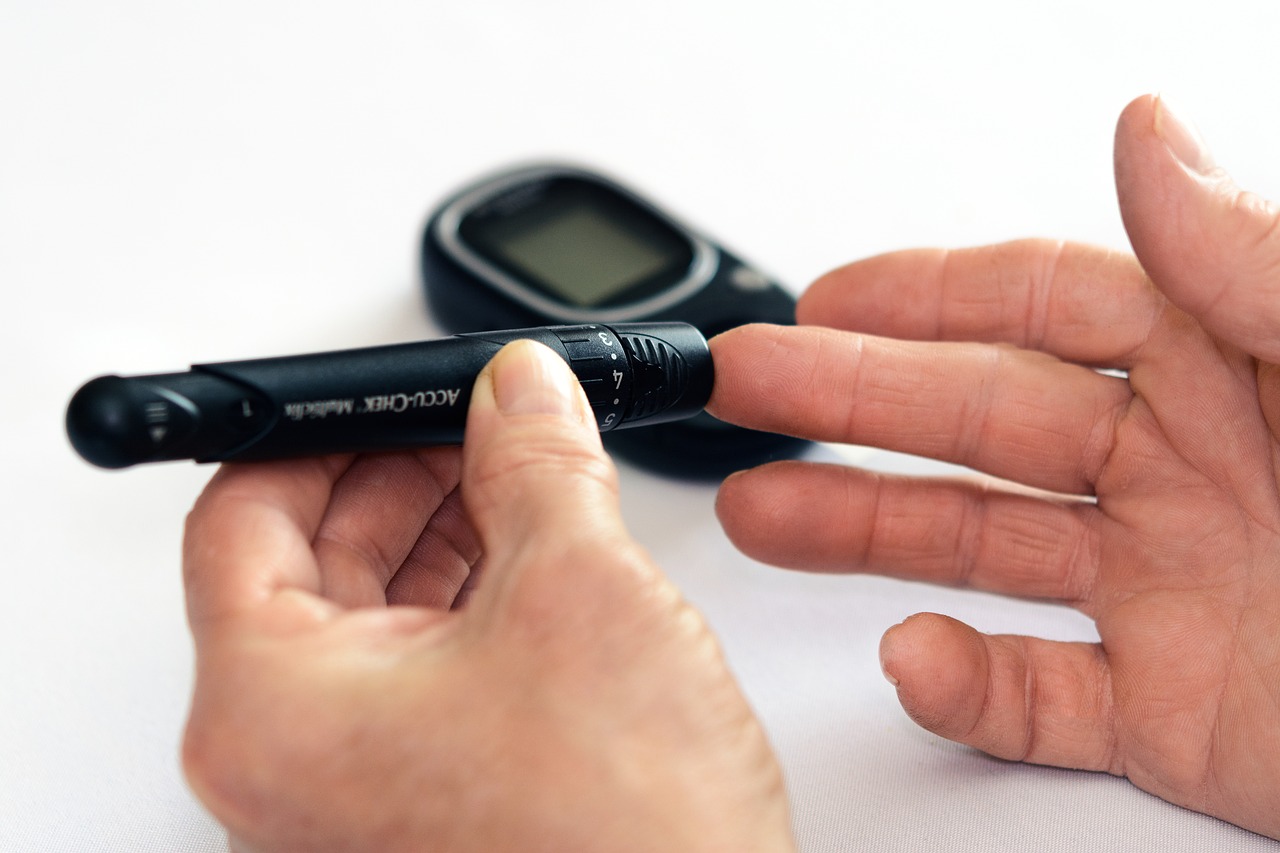
Diabetes drugs are once again under the microscope as the Food and Drug Administration (FDA) has found certain samples of sitagliptin contain nitrosamine – a possible cancer-causing ingredient – at levels above what is allowable by the agency.
Sitagliptin is used for the management of high blood sugar in individuals that have Type 2 diabetes, but it is not the first ingredient to raise a flag with the FDA over allowable levels that could cause cancer.
Several makes of the diabetes drugs metformin were previously recalled under the agency’s supervision, as well as a number of high blood pressure pills and Pfizer’s smoking cessation medication – Chantix – all due to a possible cancer-causing risk from an ingredient known as NDMA or N-Nitrosodimethylamine in levels higher than were allowed by the FDA.
Now Merck’s diabetes drugs Januvia, Janumet, and Steglujan, which contain sitagliptin with higher than allowable levels of nitrosamine, are being reviewed by the agency. The pills are still being permitted to be made to stave off a shortage of medication, Bloomberg reported.
In an email to the news outlet, Merck confirmed that it “recently detected a nitrosamine identified as NTTP in some batches of our sitagliptin-containing medicines,” adding that it is working with health officials globally to put in place quality control measures to ensure the drugs meet FDA interim limits.
The FDA said that it allows 37 nanograms per day of nitrosamine in a drug, but to avoid any shortages of the diabetes medications, it is allowing up to 246.7 nanograms daily. The agency did maintain that cancer risk with the allowable increase is minimal.
The FDA said, “It could be dangerous for patients with this condition to stop taking their sitagliptin without first talking to their health care professional.” It also “recommends prescribers continue to use sitagliptin when clinically appropriate to prevent a gap in patient treatment.”
Januvia is the third best-selling drug for Merck, followed by Janumet, with revenues of $3.3 billion and nearly $2 billion, respectively, in 2021 for the company, Bloomberg said.